New hands-on session coming soon
How to use
Audience: patient advocates, patient groups and sponsors.
Introduction to Cost Modeling & Budget Prediction for Decentralised Clinical Research
Announcing a new online workshop series on costing and budgeting decentralised clinical trials. In this first session, we will cover the following topics:
Costing decentralised clinical trials, the quick and clean way
Budget and value drivers
Study design considerations
Quantifying patient burden and recruitment benefits
Developing value for money budgets using cost prediction modeling
Audience: Pharma Clinical Development, Finance, Procurement, Trial Sites, CROs Academic researchers, Regulation/ Policy and Investors
To register for free: Email info@trialvalueapp.com and include your name, title, company details with any questions.
Introduction to Cost Modeling & Budget Prediction for Decentralised Clinical Research
Announcing a new online workshop series on costing and budgeting decentralised clinical trials. In this first session, we will cover the following topics:
Costing decentralised clinical trials, the quick and clean way
Budget and value drivers
Study design considerations
Quantifying patient burden and recruitment benefits
Developing value for money budgets using cost prediction modeling
Audience: Pharma Clinical Development, Finance, Procurement, Trial Sites, CROs Academic researchers, Regulation/ Policy and Investors
To register for free: Email info@trialvalueapp.com and include your name, title, company details with any questions.
Introduction to Cost Modeling & Budget Prediction for Decentralised Clinical Research
Announcing a new online workshop series on costing and budgeting decentralised clinical trials. In this first session, we will cover the following topics:
Costing decentralised clinical trials, the quick and clean way
Budget and value drivers
Study design considerations
Quantifying patient burden and recruitment benefits
Developing value for money budgets using cost prediction modeling
Target Audience: Pharma Clinical Development, Finance, Procurement, Trial Sites, CROs Academic researchers, Regulation/ Policy Advocates and Investors
To register for free: Email info@trialvalueapp.com and include your name, title, company details with any questions/comments that you have.
Introduction to Cost Modeling & Budget Prediction for Decentralised Clinical Research
Announcing a new online workshop series on costing and budgeting decentralised clinical trials. In this first session, we will cover the following topics:
Costing decentralised clinical trials, the quick and clean way
Budget and value drivers
Study design considerations
Quantifying patient burden and recruitment benefits
Developing value for money budgets using cost prediction modeling
Target Audience: Pharma Clinical Development, Finance, Procurement, Trial Sites, CROs Academic researchers, Regulation/ Policy Advocates and Investors
To register for free: Email info@trialvalueapp.com and include your name, title, company details with any questions/comments that you have.
Introduction to Cost Modeling & Budget Prediction for Decentralised Clinical Research
Announcing a new online workshop series on costing and budgeting decentralised clinical trials. In this first session, we will cover the following topics:
Costing decentralised clinical trials, the quick and clean way
Budget and value drivers
Study design considerations
Quantifying patient burden and recruitment benefits
Developing value for money budgets using cost prediction modeling
Target Audience: Pharma Clinical Development, Finance, Procurement, Trial Sites, CROs Academic researchers, Regulation/ Policy Advocates and Investors
To register for free: Email info@trialvalueapp.com and include your name, title, company details with any questions/comments that you have.
Introduction to Cost Modeling & Budget Prediction for Decentralised Clinical Research
Announcing a new online workshop series on costing and budgeting decentralised clinical trials. In this first session, we will cover the following topics:
Costing decentralised clinical trials, the quick and clean way
Budget and value drivers
Study design considerations
Quantifying patient burden and recruitment benefits
Developing value for money budgets using cost prediction modeling
Target Audience: Pharma Clinical Development, Finance, Procurement, Trial Sites, CROs Academic researchers, Regulation/ Policy Advocates and Investors
To register for free: Email info@trialvalueapp.com and include your name, title, company details with any questions/comments that you have.
Introduction to Cost Modeling & Budget Prediction for Decentralised Clinical Research
Announcing a new online workshop series on costing and budgeting decentralised clinical trials. In this first session, we will cover the following topics:
Costing decentralised clinical trials, the quick and clean way
Budget and value drivers
Study design considerations
Quantifying patient burden and recruitment benefits
Developing value for money budgets using cost prediction modeling
Target Audience: Pharma Clinical Development, Finance, Procurement, Trial Sites, CROs Academic researchers, Regulation/ Policy Advocates and Investors
To register for free: Email info@trialvalueapp.com and include your name, title, company details with any questions/comments that you have.
Introduction to Cost Modeling & Budget Prediction for Decentralised Clinical Research
Announcing a new online workshop series on costing and budgeting decentralised clinical trials. In this first session, we will cover the following topics:
Costing decentralised clinical trials, the quick and clean way
Budget and value drivers
Study design considerations
Quantifying patient burden and recruitment benefits
Developing value for money budgets using cost prediction modeling
Target Audience: Pharma Clinical Development, Finance, Procurement, Trial Sites, CROs Academic researchers, Regulation/ Policy Advocates and Investors
To register for free: Email info@trialvalueapp.com and include your name, title, company details with any questions/comments that you have.
Introduction to Decentralised Clinical Trial Design and Budgeting
Decentralising clinical research - hype and reality
Players and game changers
Cost and value equation
Study design considerations
Quantifying patient burden reduction
Scenario simulation
Attendees: Clinical operations, clinical research, investigator sites, outsourcing/procurement, finance, patient organisations, regulatory and policy advocates.
Financial Benchmarking in Biopharma Clinical Development - Tools and Strategies
Update: This session will address how to select the right tools and strategies for financial benchmarking in clinical development.
Audience: Clinical Development, Clinical Operations, Financial Planning and Control, Procurement, Medical Affairs, Management, Outsourcing and Compliance
Workshop details will be sent to all registered participants before the event. A minimum of 5 participants are required. Please register using the form below:
Fee:
Pharma/Biotech: EUR 99 + vat
Investigator sites, Research Centres, Non-profit: Free
CLINBIZ SUMMIT, 13 to 14 March 2019, NJ, US
Connect with top-notch industry speakers delivering impactful presentations with practical solutions and best practices in clinical outsourcing, clinical contracting and clinical financial management topics
Register now!You can’t afford to miss it.
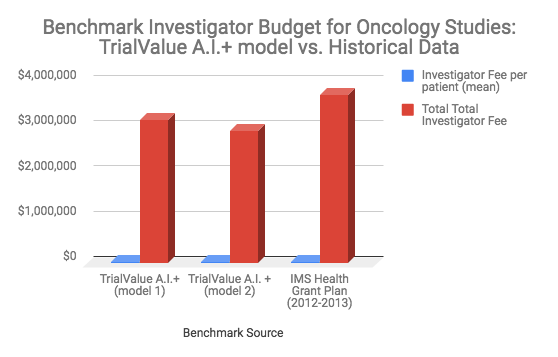
TrialValue A.I.+ : Introduction to Machine Learning enabled Clinical Research Budget Prediction and Benchmarking Model
This session will provide an overview of TrialValue A.I.+ and how the model is used at the early stage of clinical program design for financial planning, complexity analysis, budget development and cost benchmarking.
Audience: Heads of Clinical Development, Project Directors, R&D Finance, CFOs, Clinical Operations, Procurement, Program Planning, Business Development, Investors and Commercial Teams.
Additional details will be sent to all registered participants closer to the time.
Maximum number of places available: 15. Minimum number required for the session to take place is 8.
Please register using the form below:
Financial Benchmarking in Biopharma Clinical Development - Tools and Strategies - January 2019
This session will address how to select the right tools and strategies for financial benchmarking in clinical development.
Audience: Clinical Development, Finance, Medical Affairs, Management, Procurement, Outsourcing and Operations.
Meeting details will be sent to all registered participants two weeks before the event.
Please register using the form below:
TrialValue partners/user group meet-up: Barcelona
TrialValue will be hosting a Lunch and learn session on the topic of budget optimisation using human-machine learning algorithms.
Venue: AC Hotel Barcelona Forum
TrialValue A.I.+ Launch Workshop - November 2018
16.00 - Welcome and introductions
16.15 - What is TrialValue A.I.+
17.00 - How it works & Demo
17.15 - Q&A & Discussion
18.00 - Close and drinks
PCMG Annual Conference 2018, 6 - 8 June, Malta
Attending annual event and will be providing updates on TrialValue A.I - our new Machine Learning enabled benchmarking algorithm. Looking forward to the clinical outsourcing event of the year.
Budget Challenge Results to be announced
Blinded results of Budget Challenge to be announced.
CBI 10th Clinical Trial Budgeting & Forecasting Conference, 2-3 May Philadelphia
10th Annual CBI’s Clinical Trial Budgeting & Forecasting
Clinical Trial Budget Efficiency Challenge - Are you getting value for money?
Clinical research projects are costly and budget overruns are common. Teams often feel dissatisfied as their studies end and see their projects haven't delivered good value for money. What's the remedy to this "buyers' remorse"?
Take the TrialValue Budget Challenge to see how your project compares with our optimal budget benchmark, peers and the industry's best. You'll get a free budget analysis report and tips on how to extract more value from your clinical project.
How it works:
1. Register by signing up through app login
2. Enter key assumptions for a completed or active phase II or III study
3. Enter category level total costs (actual or budgeted figures) eg, site cost, biometrics, clinical operations, pass-through etc
4. Check, save and submit
5. Get a free 3 months license on registration and a report within 3 days of challenge closing date
Who can participate:
Clinical trial teams in pharmaceutical and biotech companies, CROs and academic research organizations.
Together, we can transform the clinical development enterprise.
Register Here!
Challenge starts on 20 November and runs until 20 April 2018 (period extended by 2 months). Blinded results will be published by May 30, 2018. All information provided will be treated as confidential.
Note: This research challenge is for educational purposes only.
TrialValue Platform On-line Seminar: simply a better way to do clinical development budgets
Session 2.
Audience: Biotech, clinical stage companies, small/mid-size pharma, Sites, Academic research organisations and CROs.
Functions: Clinical Research/Operations, Finance, Procurement, Business Development, Program/Project Management, Medical Affairs, and Executive management.
Together, we can add value to the clinical development enterprise.
TrialValue Platform On-line Seminar: simply a better way to do clinical development budgets - September 2017
Larry Ajuwon, Director, Rhieos-Ventures and Lead Partner at the TrialValueapp will present the platform and discuss the new approaches, practices and tools for planning and reducing the costs of clinical development projects.
Audience: Biotech, clinical stage companies, small/mid-size pharma, Sites, Academic research organisations and CROs.
Functions: Clinical Research/Operations, Finance, Procurement, Business Development, Program/Project Management, Medical Affairs, and Executive management.
Together, we can add value to the clinical development enterprise.